Halogenated organic compounds such as bromoform (CHBr₃) are common pollutants hidden in atmospheric cloud droplets and aqueous aerosols. Their photochemical reactions are directly related to ozone layer protection and air quality improvement and represent a key challenge that urgently needs to be addressed in the field of global environmental governance. These molecules exhibit a special phenomenon of atomic "roaming". However, in liquid-phase environmental systems, how solvents regulate the reaction pathway has long lacked direct experimental evidence, and the underlying mechanism has remained unclear.
Recently, Professor Lan Jinggang from the Faculty of Synthetic Biology at Shenzhen University of Advanced Technology, in collaboration with Researcher Kong Qingyu from the French SOLEIL Synchrotron Radiation Source Center and other teams, published research findings inNature Communications. Using cutting-edge technology, they precisely dissected the underlying logic of solvent-regulated ultrafast molecular reactions, bringing breakthrough progress to precise atmospheric pollution control and efficient regulation of chemical reactions.
The research team selected bromoform as a representative object, innovatively integrating femtosecond time-resolved X-ray solution scattering technology, machine learning molecular simulation, and theoretical calculation methods. By comparatively analyzing the molecular reaction patterns in two different solvents — methanol and methylcyclohexane — they ultimately confirmed that solvents are never mere "bystanders"in molecular reactions but rather the "core regulators"that determine the reaction pathway.
In polar solvents such as methanol, the bromine atoms dissociated from bromoform tend to "roam"near the molecule and actively interact with solvent molecules, directly initiating degradation reactions. In inert solvents such as methylcyclohexane, the roaming bromine atoms are more inclined to recombine with fragments of the parent molecule, forming stable intermediate isomers. In simple terms, changing the solvent can completely rewrite the reaction trajectory and final products of pollutant molecules.
The team also utilized AI-accelerated simulation technology to clearly reconstruct the atomic motion trajectories at the femtosecond timescale, making the originally invisible and intangible microscopic reaction processes observable and analyzable. This not only strengthens the credibility of the experimental conclusions but also fills a theoretical gap in the field of liquid-phase molecular roaming reactions.
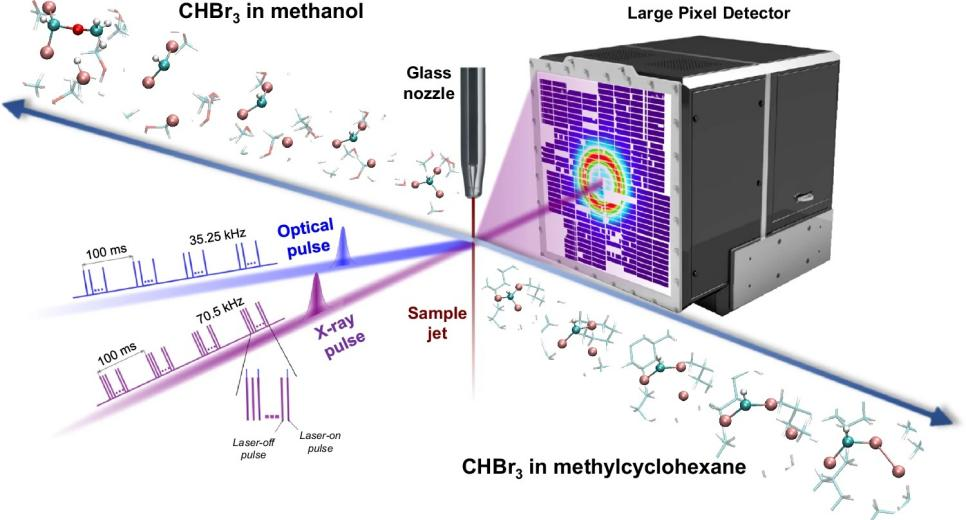
Figure 1. Schematic diagram of time-resolved X-ray solution scattering. The CHBr₃ solution is injected into methanol or methylcyclohexane solvent and ejected through a cylindrical nozzle to form a cylindrical liquid jet with a diameter of 100 micrometers. The liquid flow is excited using a femtosecond laser pulse with a wavelength of 267 nm, and the excited liquid system is probed with a time-delayed X-ray pulse.
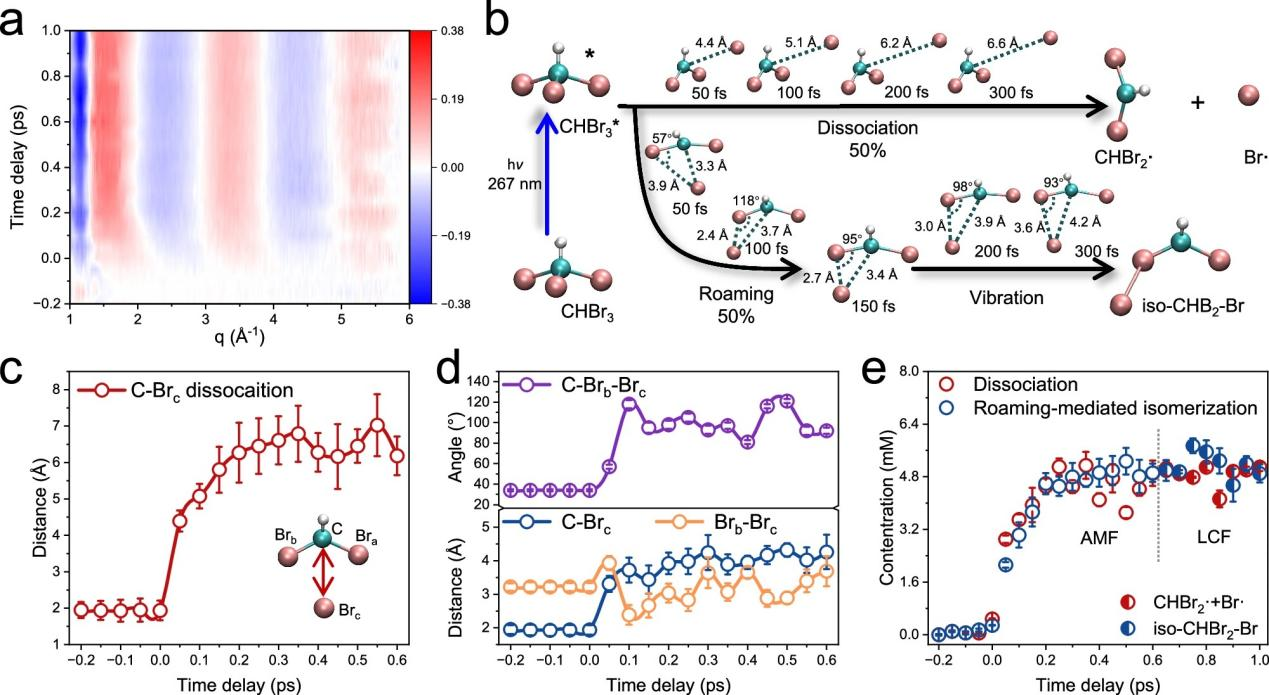
Figure 2. Photochemical reaction kinetics of chlorobromomethane in methanol
The practical value of this achievement is highly significant: on the one hand, it helps researchers gain a more thorough understanding of the transformation patterns of halogenated pollutants in the atmosphere and water bodies, providing scientific evidence for environmental protection efforts such as ozone layer repair, smog control, and water purification; on the other hand, it guides R&D personnel to precisely select solvents and controllably regulate the selectivity and product types of chemical reactions, laying a solid theoretical foundation for the development of new environmentally friendly materials and green fine chemical synthesis.
Professor Lan Jinggang from the Faculty of Synthetic Biology at Shenzhen University of Advanced Technology, Professor Weng Zuqian from the School of Physical Science and Technology at ShanghaiTech University, Dmitry Khakhulin from the European X-ray Free-Electron Laser Facility (European XFEL), and Researcher Kong Qingyu from the French SOLEIL Synchrotron Radiation Source Center are the co-corresponding authors. This project received support from the European XFEL experimental program, the National Natural Science Foundation of China, and the supercomputing resources of Shenzhen University of Advanced Technology.
Link:https://www.nature.com/articles/s41467-026-69374-4
